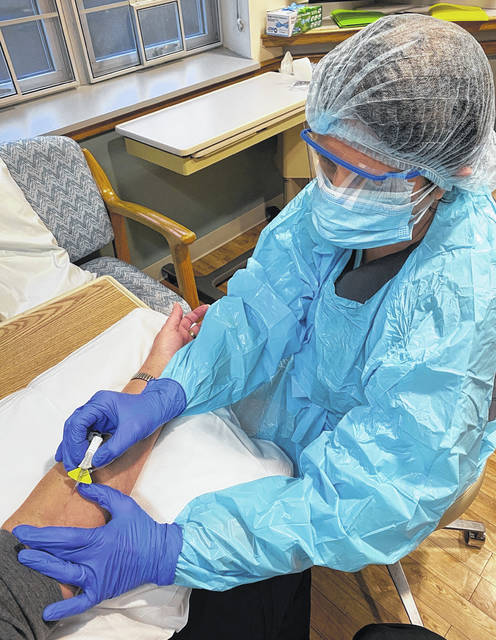
WILMINGTON – Clinton Memorial Hospital announced Wednesday that it is offering bamlanivimab and casirivimab and imdevimab — a new monoclonal antibody therapy — for treatment of non-hospitalized patients with a mild or moderate case of COVID-19.
Developed by pharmaceutical company Eli Lilly and Regeneron Pharmaceuticals, Inc., the drug recently received emergency use authorization (EUA) from the Food and Drug Administration (FDA) and is now being allocated by the U.S. Department of Health and Human Services in partnership with the Ohio Department of Health and other state agencies.
Monoclonal antibodies are laboratory-made proteins that mimic the immune system’s ability to fight off harmful viruses. Bamlanivimab and casirivimab and imdevimab are specifically designed to help block the SARS-CoV-2 virus and prevent the virus from further infecting healthy cells.
Administered intravenously, this innovative treatment is designed to help lessen the severity of COVID-19 in individuals who are COVID-19-positive and are at risk for developing a severe form of the disease.
“Clinton Memorial Hospital is proud to offer this treatment locally for qualifying patients, and our team has seen promising results in our initial patients,” said Dr. Brian Santin, Chief Medical Officer at Clinton Memorial Hospital. “Offering this treatment locally is an important step forward in helping prevent the most at-risk patients from being hospitalized due to COVID-19.”
Patients must meet specific clinical criteria, including having a lab-confirmed case of COVID-19 that is mild or moderate; having underlying health conditions or under 65 years of age; and being stable enough not to require hospitalization.
As bamlanivimab and casirivimab and imdevimab are an investigational treatment, the hospital’s supply is limited and carefully allocated by the Ohio Department of Health, and requires an order from the patient’s provider.


